On December 29, 2022, U.S. President Joe Biden signed the FDA Modernization Act 2.0, allowing researchers to use alternatives to animal testing when developing new drugs and products. This move recognises that animal models can sometimes inaccurately represent human biology and that reliable, humane alternatives exist.
One such alternative is in vitro ocular toxicity testing, a technology designed to test how substances affect the eyes without using animals. In this blog, we’ll explore the key aspects of this emerging technology and its applications in improving eye health and product safety.
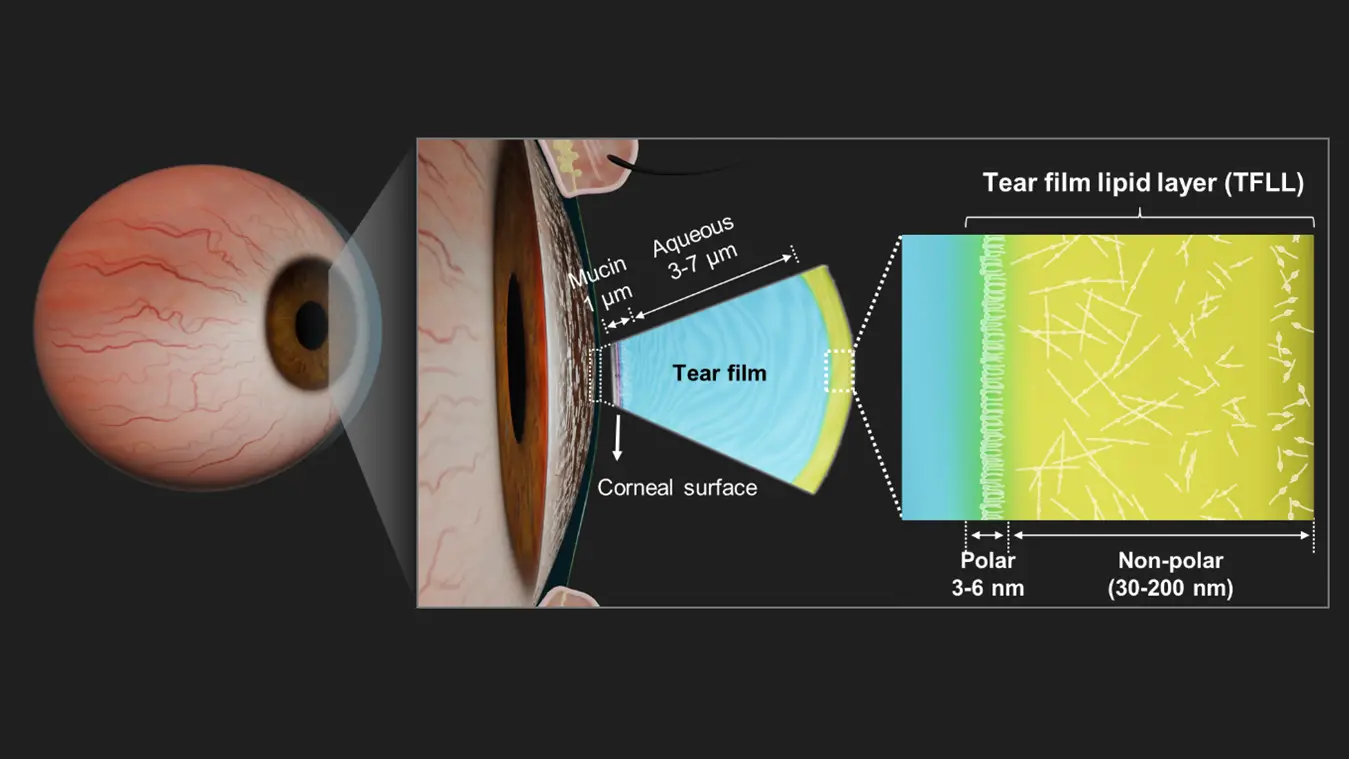
The Role of the Eye’s Tear Film
To understand the importance of ocular toxicity testing, it’s helpful to know how the eye protects itself. The eye’s surface is shielded by the Tear Film Lipid Layer (TFLL), a crucial barrier that provides lubrication, protects against microbes and maintains optical clarity. This layer consists of:
- A polar sub-layer, which is thin and interacts with the tear’s aqueous components.
- A non-polar lipid layer, which is thicker and exposed to the environment.
Together, these layers ensure the eye remains hydrated and protected from irritants. Disrupting this tear film, whether through environmental factors or harmful product ingredients, can lead to conditions like dry eye disease or infections. As a result, testing how various substances impact this delicate layer is crucial before products reach consumers.
Current Ocular Toxicity Testing Methods
Historically, the Draize rabbit eye test has been the industry standard for testing eye irritancy. This in vivo test involves applying substances to the eyes of rabbits to observe irritation or damage. While effective in some ways, it has its limitations, including ethical concerns and differences in human and animal biology that can affect the accuracy of results.
In recent years, several alternatives to animal testing have emerged:
- Organotypic models that simulate the full thickness of the cornea.
- Reconstructed human cornea assays that use human-derived cells.
- Cytotoxicity assays that assess the effect of chemicals on cultured eye cells.
Although these methods offer promising alternatives, none fully replicate the complexity of the eye’s tear film.
Introducing an In Vitro Ocular Toxicity Testing Platform
A new platform from a team of innovators from IDMxS represents a significant advancement in ocular toxicity testing. Unlike other in vitro models, it recreates both the polar and non-polar layers of the eye’s tear film, offering a more accurate representation of how substances interact with the ocular surface.
Key improvements include:
- Full representation of tear film layers: Unlike liposomal assays that fail to mimic the entire structure of the tear film, the platform includes both the polar and non-polar lipid layers, closely simulating the natural environment of the eye.
- High-throughput capabilities: The system supports parallel testing, allowing for multiple substances to be tested simultaneously. This significantly speeds up the testing process and reduces costs.
These improvements make the platform highly effective in assessing how different products, whether solid, liquid, or gas, impact the eye. As a result, it offers a more reliable and humane alternative to traditional animal testing methods.
Applications Across Various Industries

This innovative in vitro platform has broad applications in several industries:
- Lotions and Cosmetics: Cosmetic products must undergo rigorous safety testing, especially those used near the eyes. The platform allows companies to evaluate these products without the ethical concerns of animal testing, offering fast, reliable results during the formulation process.
- Artificial Tears: For individuals with dry eye disease, artificial tears are an essential part of daily life. The platform enables testing these products under various environmental conditions, ensuring they remain stable and effective in multiple settings, from dry climates to areas with high pollution.
- Contact Lens Products: Contact lens wearers need materials and solutions that are both safe and comfortable. With the IDMxS platform, manufacturers can test how different lens materials interact with the tear film, ensuring users’ optimal comfort and long-term safety.
- Pharmaceuticals Formulations: Therapeutic formulations may cause eye irritation that affect the stability of the tear film. The platform enables optimisation of the formulations to minimise potential side effects on tear film.
The Expanding Market for Ocular Health Solutions
The demand for eye care products is on the rise. According to the World Report on Vision, at least 1 billion people worldwide have some form of vision impairment. With many cases being preventable or treatable, the demand for ocular health products is projected to grow significantly. The steady growth of the global ophthalmology market drives the need for faster, more reliable, and more ethical testing methods, especially in high-demand areas like artificial tears, ocular injectables, and cosmetics.
The in vitro ocular testing market is also rapidly expanding, with a projected value beyond $1 billion by 2030. The end users of ocular toxicity testing cover various industries including personal care product, research institution, chemical, and pharmaceutical and biotechnology, highlighting its wide application.
Looking Ahead: The Future of Ocular Toxicity Testing
In vitro, ocular toxicity testing is revolutionising how we ensure the safety of products that come into contact with our eyes. With advancements like the IDMxS platform, we can now accurately test substances in a way that mimics the human eye, reducing the need for animal testing while improving the reliability of results.
The future of ocular health is bright, with safer, more ethical testing methods leading the way in product development. Get in touch with Dr James Ho (james.hcs@ntu.edu.sg) to learn more about the platform.